Fluid/Electrolyte Balance
Fluid Balance- The amount of water gained each day equals the amount lost
Electrolyte Balance - The ions gained each day equals the ions lost
Acid-Base Balance - Hydrogen ion (H+) gain is offset by their loss
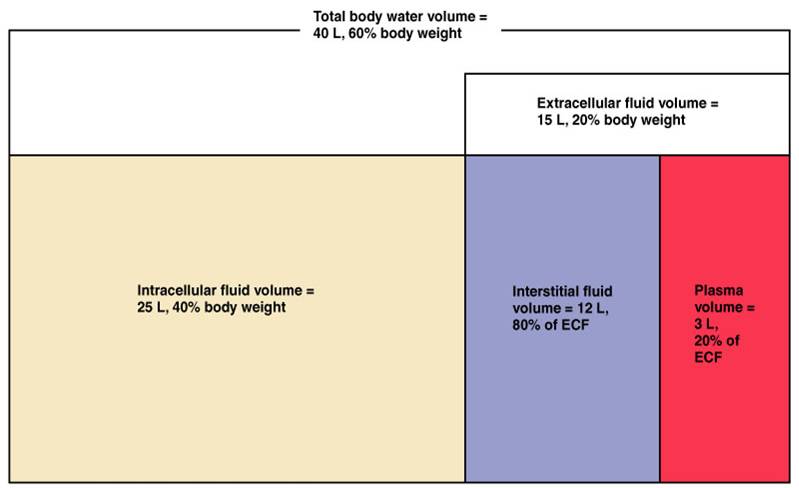
Body Fluids Compartments
- Intracellular Fluid (ICF) - fluid found in the cells (cytoplasm, nucleoplasm) comprises 60% of all body fluids.
- Extracellular Fluid (ECF) - all fluids found outside the cells, comprises 40% of all body fluids
- Interstitial Fluid - 80% of ECF is found in localized areas: lymph, cerebrospinal fluid, synovial fluid, aqueous
humor and vitreous body of eyes, between serous and visceral membranes, glomerular filtrate of kidneys.
- Blood Plasma - 20% of ECF found in circulatory system
Composition of Body Fluids
- Water is the main component of all body fluids making up 45-75% of the total body weight.
- Sources of water include
- Ingested foods and liquids (preformed water)
- Metabolic water produced during dehydration synthesis of anabolism.
- Solutes are broadly classified into
- Electrolytes are inorganic salts, all acids and bases, and some proteins
- Nonelectrolytes – examples include glucose, lipids, creatinine, and urea
- Electrolytes have greater osmotic power than nonelectrolytes
- Water moves according to osmotic gradients
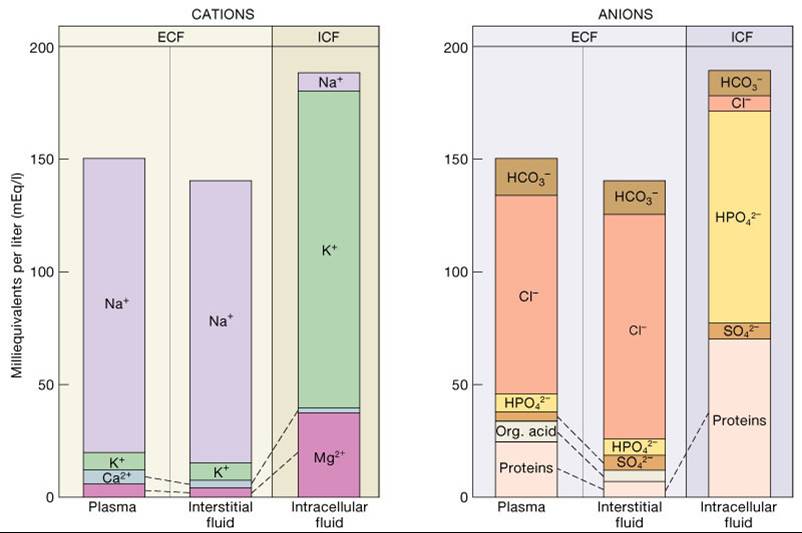
Electrolyte Composition of Body Fluids
Each fluid compartment of the body has a distinctive pattern of electrolytes
- Extracellular Fluids
- ECFs are similar except for the high protein content of plasma
- Sodium (Na+) is the major cation
- Chloride (Cl-)is the major anion
- Intracellular Fluids
- Have low sodium and chloride
- Potassium (K+) is the chief cation
- Phosphate (PO4-) is the chief anion

Extracellular and Intracellular Fluids
- Sodium and potassium concentrations in extra- and intracellular fluids are nearly opposites
- This reflects the activity of cellular ATP-dependent sodium-potassium pumps
- Electrolytes determine the chemical and physical reactions of fluids
- Ion fluxes are restricted and move selectively by active transport
- Nutrients, respiratory gases, and wastes move unidirectionally
- Plasma is the only fluid that circulates throughout the body and links external and internal environments
- Osmolalities of all body fluids are equal; changes in solute concentrations are quickly followed by osmotic changes
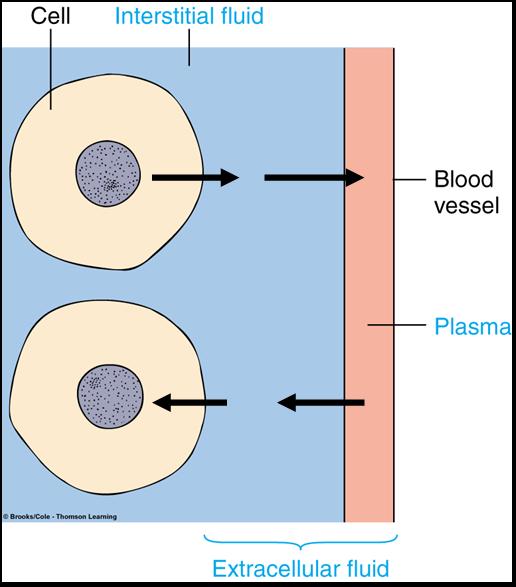
Fluid Movement Among Compartments
- Compartmental exchange is regulated by osmotic and hydrostatic pressures
- Net leakage of fluid from the blood is picked up by lymphatic vessels and returned to the bloodstream
- Exchanges between interstitial and intracellular fluids due to the selective permeability of the cellular membranes
Fluid Shifts
- If ECF becomes hypertonic relative to ICF, water moves from ICF to ECF
- If ECF becomes hypotonic relative to ICF, water moves from ECF into cells
|
 |
Regulation of Fluids And Electrolytes
- Homeostatic mechanisms respond to changes in ECF
- No receptors directly monitor fluid or electrolyte balance
- Response is to changes in plasma volume or osmotic concentrations
- All water moves passively in response to osmotic gradients
- Body content of water or electrolytes rises if intake exceeds outflow
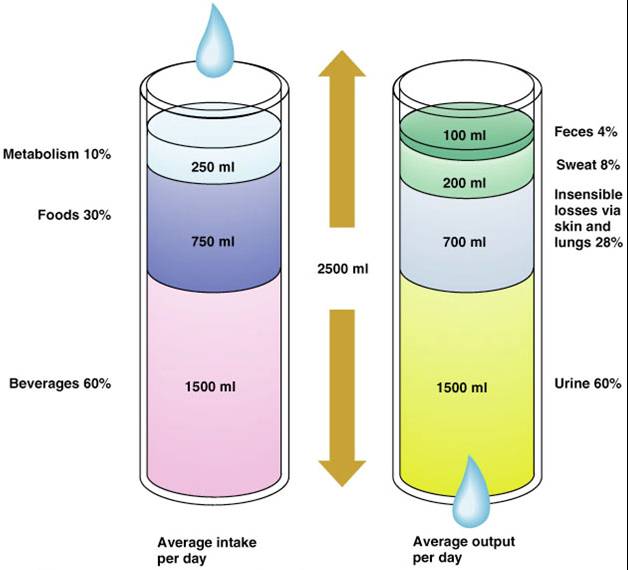
Water Balance and ECF Osmolality
- To remain properly hydrated, water intake must equal water output
- Water intake sources
- Ingested fluid (60%) and solid food (30%)
- Metabolic water or water of oxidation (10%)
Water Output
- Urine (60%)
- Feces (4%)
- Insensible losses through the skin and lungs (28%)
- Sweat (8%)
- Increases in plasma osmolality trigger thirst and release of antidiuretic hormone (ADH)
|
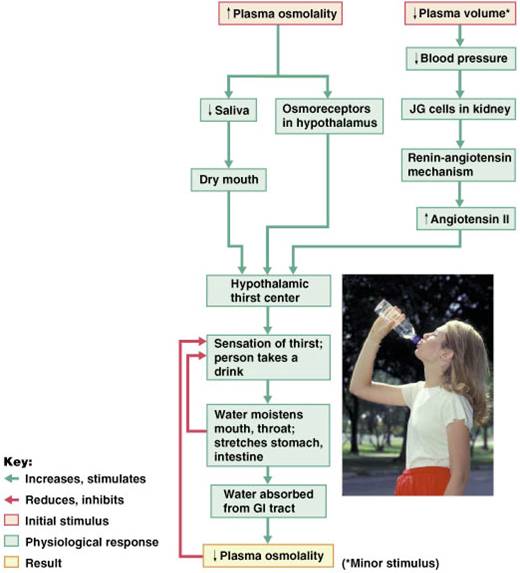
Regulation of Water Intake
- The hypothalamic thirst center is stimulated by:
- Decline in plasma volume of 10%–15%
- Increases in plasma osmolality of 1–2%
- Baroreceptor input, angiotensin II, and other stimuli
- Thirst is quenched as soon as we begin to drink water
- Feedback signals that inhibit the thirst centers include:
- Moistening of the mucosa of the mouth and throat
- Activation of stomach and intestinal stretch receptors
|
 |
Regulation of Water Output
Obligatory water losses include:
- Insensible water losses from lungs and skin
- Water that accompanies undigested food residues in feces
Obligatory water loss reflects the fact that:
- Kidneys excrete 900-1200 mOsm of solutes to maintain blood homeostasis
- Urine solutes must be flushed out of the body in water
Primary Regulatory Hormones
1. Antidiuretic hormone (ADH) (also called vasopressin)
- Is a hormone made by the hypothalamus, and stored and released in the posterior pituitary gland
- Primary function of ADH is to decrease the amount of water lost at the kidneys (conserve water), which reduces the concentration of electrolytes
- ADH also causes the constriction of peripheral blood vessels, which helps to increase blood pressure
- ADH is released in response to such stimuli as a rise in the concentration of electrolytes in the blood or a fall in blood volume or pressure. These stimuli occur when a person sweats excessively or is dehydrated.
1. Sweating or dehydration increases the blood osmotic pressure.
2. The increase in osmotic pressure is detected by osmoreceptors within the hypothalamus that constantly monitor the osmolarity ("saltiness") of the blood
3. Osmoreceptors stimulate groups of neurons within the hypothalamus to release ADH from the posterior pituitary gland.
4. ADH travels through the bloodstream to its target organs:
a. ADH tavels to the collecting tubules in the kidneys and makes the membrane more permeable to water (that is it increases water reabsorption) which leads to a decrease in urine output.
b. ADH also travels to the sweat glands where it stimulates them to decrease perspiration to conserve water.
c. ADH travels to the arterioles, where it causes the smooth muscle in the wall of the arterioles to constrict. This narrows the diameter of the arterioles which increases blood pressure.
Alcohol inhibits the production of ADH which is one of the reasons a person has increased fluid excretion after drinking alcohol!
Click here for an animation on the release of ADH in response to decreased blood volume. The animation is followed by practice questions.
2. Aldosterone -
- Is a hormone made by cells in the adrenal cortex (zona glomerulosa)
- Controls the levels of Na+ and K+ ions in extracellular fluids such as the blood
- Net result of its action is to reabsorb Na+ ions into the blood and simultaneously excrete K+ ions into the urine; because "water follows the ions," as Na+ is reabsorbed, water is also reabsorbed.
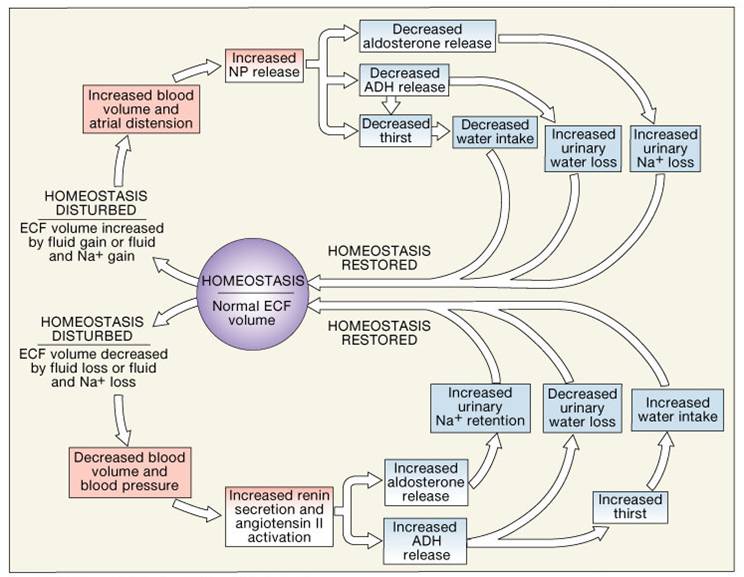
3. Natriuretic Peptides (Atrial Natriuretic Peptide and Brain Natriuretic Peptide)
- Atrial natriuretic peptide (ANP) is a hormone made by cells in the right atrium whenever blood volume increases (atria are stretched)
- Brain natriuretic peptide (BNP) is a hormone made by cells in the ventricles in response to excessive stretching of the ventricles
- In general, the effects of ANP and BNP are opposite to those of angiotensin II
- Both ANP and BNP promote the loss of sodium ions and water at the kidneys in the urine, inhibit rennin release, and inhibit the secretion of ADH and aldosterone
- By inducing blood vessels to dilate and water to be excreted in the urine, ANP and BNP reduce both blood volume and blood pressure
Disorders of Water Balance
Dehydration:
- Water loss exceeds water intake and the body is in negative fluid balance
- Causes include: hemorrhage, severe burns, prolonged vomiting or diarrhea, profuse sweating, water deprivation, and diuretic abuse
- Signs and symptoms: cottonmouth, thirst, dry flushed skin, and oliguria (decreased production of urine)
Hypotonic Hydration:
- Renal insufficiency or an extraordinary amount of water ingested quickly can lead to cellular overhydration, or water intoxication
- ECF is diluted – sodium content is normal but excess water is present resulting hyponatremia promotes net osmosis into tissue cells
- These events must be quickly reversed to prevent severe metabolic disturbances, particularly in neurons
Electrolyte Balance
- Electrolytes are salts, acids, and bases, but electrolyte balance usually refers only to salt balance
- Salts are important for:
- Essential minerals
- Controls osmosis between fluid compartments
- Help maintain acid-base balance
- Carry electrical (ionic) current for action potentials
Sodium in Fluid and Electrolyte Balance
- Sodium holds a central position in fluid and electrolyte balance
- Sodium is the single most abundant cation in the ECF
- Accounts for 90-95% of all solutes in the ECF
- Contribute 280 mOsm of the total 300 mOsm ECF solute concentration
- The role of sodium in controlling ECF volume and water distribution in the body is a result of:
- Sodium being the only cation to exert significant osmotic pressure
- Sodium ions leaking into cells and being pumped out against their electrochemical gradient
- Sodium concentration in the ECF normally remains stable
- Rate of sodium uptake across digestive tract directly proportional to dietary intake
- Sodium losses occur through urine and perspiration
- Changes in plasma sodium levels affect:
- Plasma volume, blood pressure
- ICF and interstitial fluid volume
- Large variations in sodium are corrected by homeostatic mechanisms
- If sodium levels are too low, antidiuretic hormone (ADH) and aldosterone are secreted
- If sodium levels are too high, atrial natriuretic peptide (ANP) is secreted
|
Sodium balance
|
|
Regulation of Sodium Balance: Aldosterone
- A decrease in Na+ levels in the plasma stimulates aldosterone release
- The kidneys detect the decrease in Na+ levels and cause a series of reactions referred to as the renin-angiotensin-aldosterone mechanisms.
- This is mediated by the juxtaglomerular apparatus, which releases renin in response to:
- Sympathetic nervous system stimulation
- Decreased filtrate osmolality
- Decreased stretch (due to decreased blood pressure)
- Sodium reabsorption
- 65% of sodium in filtrate is reabsorbed in the proximal tubules
- 25% is reclaimed in the loops of Henle
- When aldosterone levels are high, all remaining Na+ is actively reabsorbed
- Water follows sodium if tubule permeability has been increased with ADH
Atrial Natriuretic Hormone (ANH)
- Is released in the heart atria as a response to stretch (elevated blood pressure),
- It has potent diuretic and natriuretic effects
- It promotes excretion of sodium and water, inhibits angiotensin II production
Potassium Balance
- Potassium ion concentrations in ECF are low
- Not as closely regulated as sodium
- Potassium ion excretion increases as ECF concentrations rise, aldosterone secreted, pH rises
- Potassium retention occurs when pH falls

Regulation of Potassium Balance
- Relative ICF-ECF potassium ion concentration affects a cell’s resting membrane potential
- Potassium controls its own ECF concentration via feedback regulation of aldosterone release
- An increase in K+ levels stimulates the release of aldosterone through the renin-angiotensin-aldosterone mechanism or through the direct release of aldosterone from the adrenal cortex cells
- Aldosterone stimulates potassium ion excretion from the kidneys
- In cortical collecting ducts, for each Na+ reabsorbed, a K+ is excreted
- When K+ levels are low, the amount of secretion and excretion is kept to a minimum
- Excessive ECF potassium (hyperkalemia) decreases membrane potential
- Too little potassium (hypokalemia) causes hyperpolarization and nonresponsiveness
- Hyperkalemia and hypokalemia can
- Disrupt electrical conduction in the heart
- Lead to sudden death
- Hydrogen ions shift in and out of cells lead to corresponding shifts in potassium in the opposite direction and interferes with activity of excitable cells
Regulation of Calcium
- Ionic calcium in ECF is important for blood clotting, cell membrane permeability, and secretory behavior
- Hypocalcemia increases excitability and causes muscle tetany
- Hypercalcemia inhibits neurons and muscle cells and may cause heart arrhythmias
- Two hormones regulate blood calcium levels:
- Parathyroid Hormone (PTH) (made by the parathyroid glands)
- Calcitonin (CT) (made by the thyroid glands)
- As calcium-rich foods are ingested, blood calcium levels rise. The thyroid gland releases calcitonin (CT) .
- CT binds to receptors on osteoblasts (bone-forming cells).
- This triggers the osteoblasts to deposit calcium salts into bone throughout the skeletal system.
- This causes the blood calcium levels to fall.
- CT stops being produced when blood calcium levels return to normal.
- When blood calcium levels fall, the parathyroid glands (located on posterior surface of the thyroid gland) release PTH.
- PTH binds to receptors on osteoclasts (bone-degrading cells) within the skeletal system
- The osteoclasts decompose bone and release calcium into the blood.
- The blood calcium level rises
- PTH stops being produced when blood calcium levels return to normal.
- - Calcium reabsorption and phosphate excretion go hand in hand
- Filtered phosphate is actively reabsorbed in the proximal tubules
- In the absence of PTH, phosphate reabsorption is regulated by its transport maximum and excesses are excreted in urine
- High or normal ECF calcium levels inhibit PTH secretion
- Release of calcium from bone is inhibited
- Larger amounts of calcium are lost in feces and urine
- More phosphate is retained
Regulation of Anions
- Chloride (Cl-) is the major anion accompanying sodium in the ECF
- 99% of chloride is reabsorbed under normal pH conditions
- When acidosis occurs, fewer chloride ions are reabsorbed
- Other anions have transport maximums and excesses are excreted in urine
Acid-Base Balance
- Molecules that are dissolved in water may dissociate into charged ions.
- An acid is a substance that increases the number of H+ ions in a solution.
- A base is a substance that decreases the number of H+ ions in a solution.
- Normal pH of body fluids:
- Arterial blood is 7.4
- Venous blood and interstitial fluid is 7.35
- Intracellular fluid is 7.0
- Important part of homeostasis because cellular metabolism depends on enzymes, and enzymes are sensitive to pH.
- Challenges to acid-base balance due to cellular metabolism: produces acids – hydrogen ion donors
- Acidosis (physiological acidosis) is a blood pH below 7.35. Its principal effect is depression of the central nervous system by depressing synaptic transmissions
- Alkalosis is a blood pH above 7.45. Its principal effect is overexcitability of the central nervous system through facilitation of synaptic transmission
Sources of Hydrogen Ions
Most hydrogen ions originate from cellular metabolism
- Breakdown of phospho rus-containing proteins releases phosphoric acid into the ECF
- Anaerobic respiration of glucose produces lactic acid
- Fat metabolism yields organic acids and ketone bodies
- Transporting carbon dioxide as bicarbonate releases hydrogen ions
Hydrogen Ion Regulation
Concentration of hydrogen ions is regulated sequentially by:
- Chemical buffer systems act within seconds
- The respiratory center in the brain stem acts within 1-3 minutes
- Renal mechanisms require hours to days to affect pH changes
Chemical Buffer Systems
- A buffer is a solution whose function is to minimize the change in pH when a base or an acid is added to the solution
- Most buffers consist of a weak acid (which releases H+ ions) and a weak base (which binds H+ ions)
- If an acidic solution is added to a buffer solution, the buffer will combine with the extra H+ ions and help to maintain the pH
- If a basic solution is added to a buffer solution, the buffer will release H+ ions to help maintain the pH
- There are many different buffers and each one stabilizes the pH of the solution within a specific pH range
- One buffer may be effective within a range of pH 2 to pH 6, while another buffer may be effective within a range of pH 10 to pH 12
- Strong Acids – all their H+ is dissociated completely in water
Weak Acids – dissociate partially in water and are efficient at preventing pH changes
Strong Bases – dissociate easily in water and quickly tie up H+
Weak Bases – accept H+ more slowly (e.g., HCO3¯ and NH3)
The three main buffer systems in our bodies are the:
- bicarbonate buffer system
- phosphate buffer system
- protein buffer system
|
|
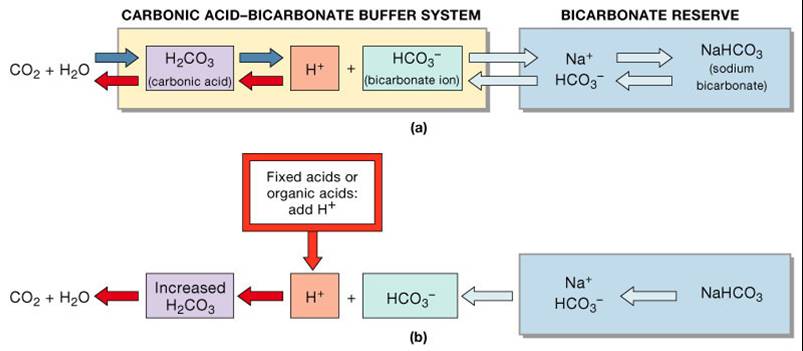
1. Bicarbonate Buffer System
- Is a mixture of carbonic acid (H2CO3) and its salt, sodium bicarbonate (NaHCO3) (potassium or magnesium bicarbonates
- If strong acid is added:
- Hydrogen ions released combine with the bicarbonate ions and form carbonic acid (a weak acid)
- The pH of the solution decreases only slightly
- If strong base is added:
- It reacts with the carbonic acid to form sodium bicarbonate (a weak base)
- The pH of the solution rises only slightly
- This system is the only important ECF buffer
2. Phosphate Buffer System
- Nearly identical to the bicarbonate system
- Its components are:
- Sodium salts of dihydrogen phosphate (NaH2PO4¯), a weak acid
- Monohydrogen phosphate (Na2HPO42¯), a weak base
- This system is an effective buffer in urine and intracellular fluid
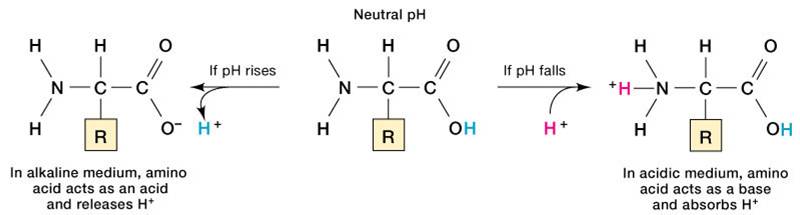
3. Protein Buffer System
- Plasma and intracellular proteins are the body’s most plentiful and powerful buffers
- Some amino acids of proteins have:
- Free organic acid groups (weak acids)
- Groups that act as weak bases (e.g., amino groups)
- Amphoteric molecules are protein molecules that can function as both a weak acid and a weak base
Physiological Buffer Systems
- The respiratory system regulation of acid-base balance is a physiological buffering system
- There is a reversible equilibrium between:
- Dissolved carbon dioxide and water
- Carbonic acid and the hydrogen and bicarbonate ions
- CO2 + H2O « H2CO3 « H+ + HCO3¯
- When hypercapnia or rising plasma H+ occurs:
- Deeper and more rapid breathing expels more carbon dioxide
- Hydrogen ion concentration is reduced
- Alkalosis causes slower, more shallow breathing, causing H+ to increase
- Respiratory system impairment causes acid-base imbalance (respiratory acidosis or respiratory alkalosis)
Renal Mechanisms of Acid-Base Balance
- Chemical buffers can tie up excess acids or bases, but they cannot eliminate them from the body
- The lungs can eliminate carbonic acid by eliminating carbon dioxide
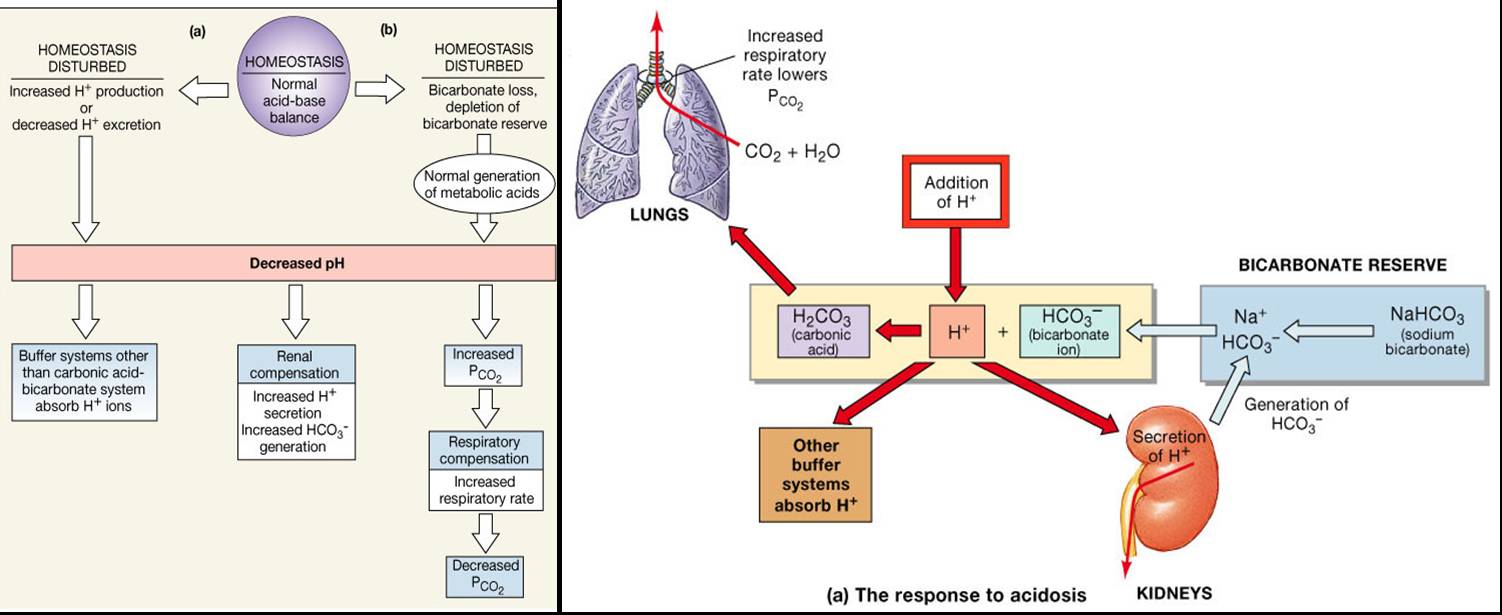
- Only the kidneys can rid the body of metabolic acids (phosphoric, uric, and lactic acids and ketones) and prevent metabolic acidosis
- The ultimate acid-base regulatory organs are the kidneys
- The most important renal mechanisms for regulating acid-base balance are conserving (reabsorbing) or generating new bicarbonate ions and excreting bicarbonate ions
- Losing a bicarbonate ion is the same as gaining a hydrogen ion; reabsorbing a bicarbonate ion is the same as losing a hydrogen ion
Reabsorption of Bicarbonate
- Carbonic acid formed in filtrate dissociates to release carbon dioxide and water
- Carbon dioxide then diffuses into tubule cells, where it acts to trigger further hydrogen ion secretion
- For each hydrogen ion secreted, a sodium ion and a bicarbonate ion are reabsorbed by the PCT cells
- Secreted hydrogen ions form carbonic acid
- Thus, bicarbonate disappears from filtrate at the same rate that it enters the peritubular capillary blood
Generating New Bicarbonate Ions
- Two mechanisms carried out by tubule cells generate new bicarbonate ions
- Both involve renal excretion of acid via secretion and excretion of hydrogen ions or ammonium ions (NH4+)
Hydrogen Ion Excretion
- In response to acidosis hydrogen ions must be counteracted by generating new bicarbonate
- Kidneys generate bicarbonate ions and add them to the blood
- An equal amount of hydrogen ions are added to the urine
- Dietary The excreted hydrogen ions must bind to buffers (phosphate buffer system) in the urine and excreted
- Bicarbonate generated is:
- Moved into the interstitial space via a cotransport system
- Passively moved into the peritubular capillary blood
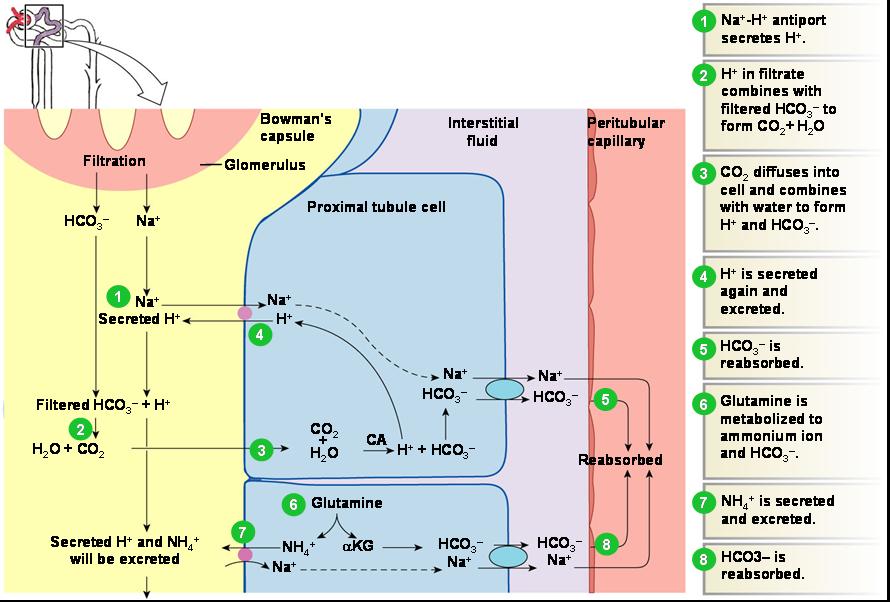
Ammonium Ion Excretion
- This method uses ammonium ions produced by the metabolism of glutamine in PCT cells
- Each glutamine metabolized produces two ammonium ions and two bicarbonate ions
- Bicarbonate moves to the blood and ammonium ions are excreted in urine
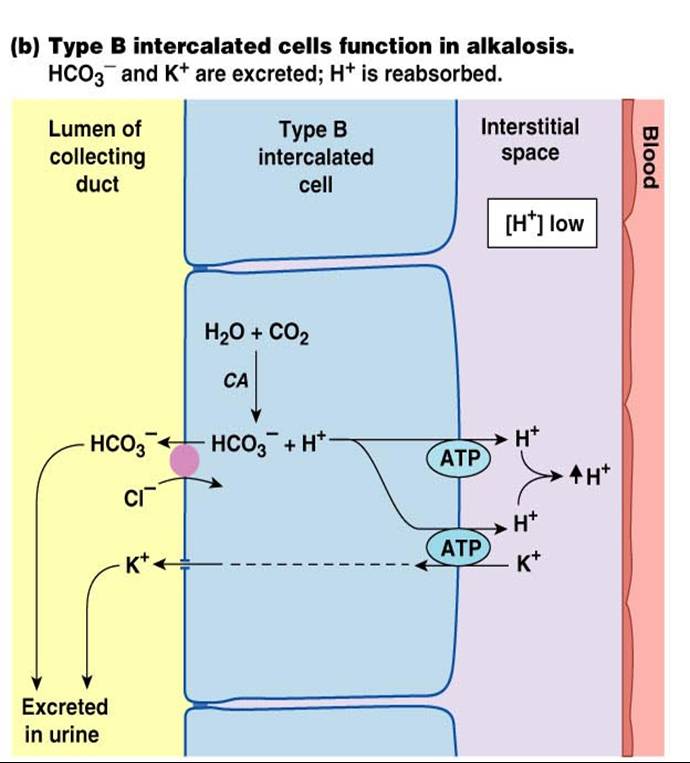
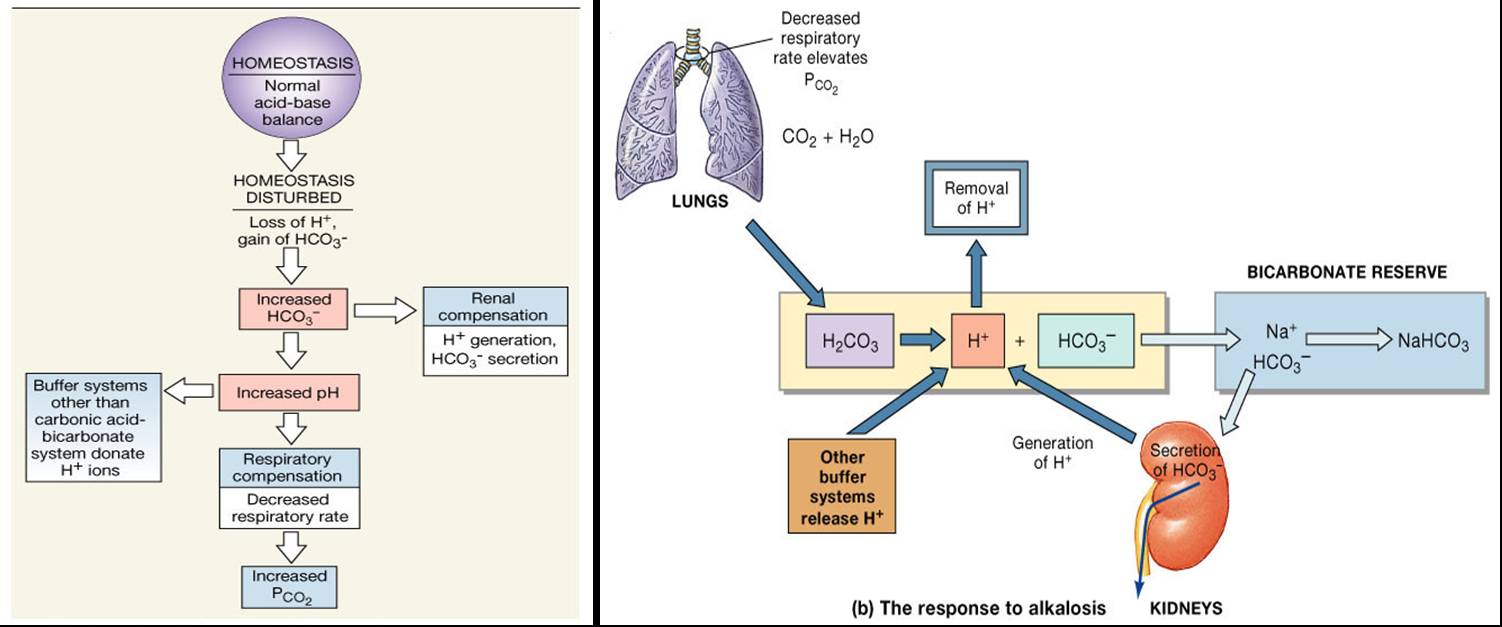
Bicarbonate Ion Secretion
- When the body is in alkalosis, tubular cells: Secrete bicarbonate ions and reclaim hydrogen ions and acidify the blood
- The mechanism is the opposite of bicarbonate ion reabsorption process
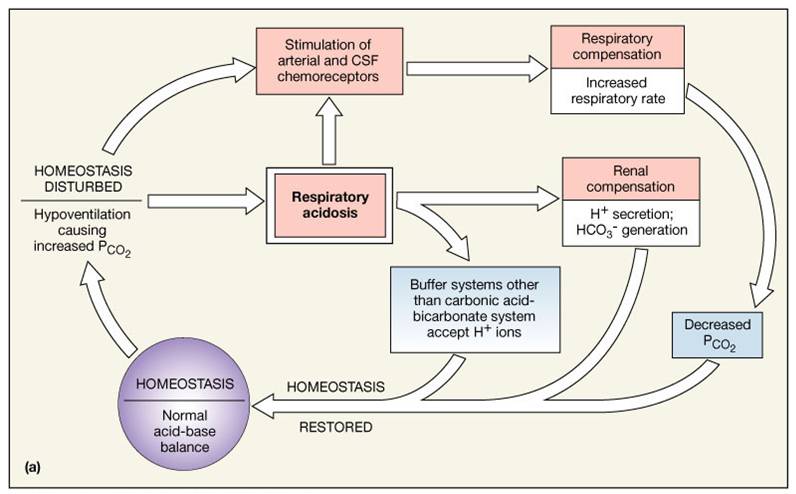
Respiratory Acidosis and Alkalosis
- Result from failure of the respiratory system to balance pH
- PCO2 is the single most important indicator of respiratory inadequacy
- PCO2 levels
- Normal PCO2 fluctuates between 35 and 45 mm Hg
- Values above 45 mm Hg signal respiratory acidosis
- Values below 35 mm Hg indicate respiratory alkalosis
Respiratory Acid-Base Regulation
- Respiratory acidosis is the most common cause of acid-base imbalance
- Occurs when a person breathes shallowly, or gas exchange is hampered by diseases such as pneumonia, cystic fibrosis, or emphysema
- Respiratory alkalosis is a common result of hyperventilation
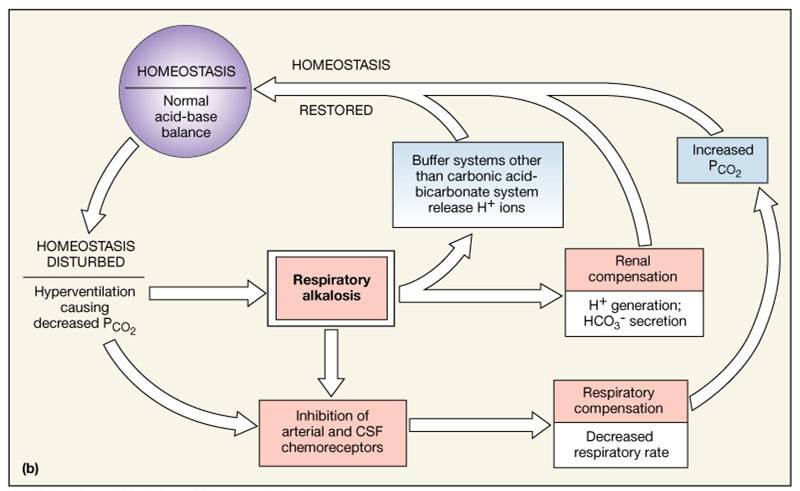
Metabolic pH Imbalance
- Metabolic acidosis is the second most common cause of acid-base imbalance.
- Typical causes are:
- Ingestion of too much alcohol and excessive loss of bicarbonate ions
- Other causes include accumulation of lactic acid, shock, ketosis in diabetic crisis, starvation, and kidney failure
- -Metabolic alkalosis due to a rise in blood pH and bicarbonate levels.
Typical causes are:
- Vomiting of the acid contents of the stomach
- Intake of excess base (e.g., from antacids)
- Constipation, in which excessive bicarbonate is reabsorbed
Respiratory/Renal Compensation/Metabolic Acidosis
- Rate and depth of breathing are elevated
- As carbon dioxide is eliminated by the respiratory system, PCO2 falls below normal
- Kidneys secrete H+ and retain/generate bicarbonate to offset the acidosis
Metabolic Alkalosis
- Pulmonary ventilation is slow and shallow allowing carbon dioxide to accumulate in the blood
- Kidneys generate H+ and eliminate bicarbonate from the body by secretion
Acid-base imbalance due to inadequacy of a physiological buffer system is compensated for by the other system.
- The respiratory system will attempt to correct metabolic acid-base imbalances
- The kidneys will work to correct imbalances caused by respiratory disease
TOP........... Main Page |
|



