|
http://www.cartage.org.lb/en/themes/sciences/astronomy/Modenastronomy/Interactionoflight/AtomicAbsorption/AtomicAbsorption.htm |
||||||||||
|
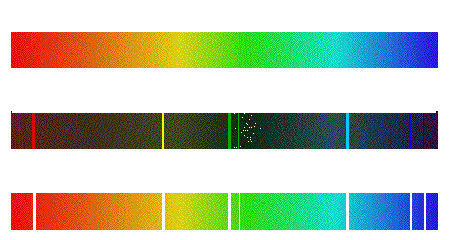
Continuum, Emission, and Absorption Spectra The corresponding spectrum may exhibit a continuum, or may have
superposed on the continuum bright lines (an emission spectrum) or dark lines (an absorption spectrum), as illustrated in the following
figure.
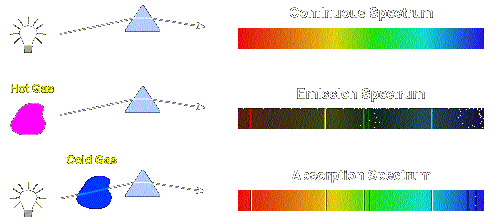
Origin of Continuum, Emission, and Absorption Spectra The origins of these three types of spectra are illustrated in
the following figure.
A continuum spectrum results when the gas pressures are
higher, so that lines are broadened by collisions between the atoms until
they are smeared into a continuum. We may view a continuum spectrum as an
emission spectrum in which the lines overlap with each other and can no
longer be distinguished as individual emission lines. An absorption spectrum occurs when light passes through a
cold, dilute gas and atoms in the gas absorb at characteristic frequencies;
since the re-emitted light is unlikely to be emitted in the same direction as
the absorbed photon, this gives rise to dark lines (absence of light) in the
spectrum. Hydrogen Emission and Absorption Series
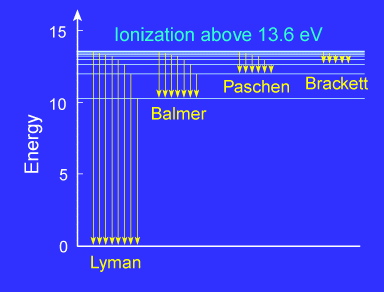
The spectrum of hydrogen is particularly important in astronomy
because most of the Universe is made of hydrogen. Emission or absorption
processes in hydrogen give rise to series, which are
sequences of lines corresponding to atomic transitions, each ending or
beginning with the same atomic state in hydrogen. Thus, for example, the Balmer Series involves transitions starting (for absorption) or ending
(for emission) with the first excited state of hydrogen, while the Lyman Series involves transitions that start or end with the ground
state of hydrogen; the adjacent image illustrates the atomic transitions that
produce these two series in emission. Because of
the details of hydrogen's atomic structure, the Balmer
Series is in the visible spectrum and the Lyman Series is in the the UV. The following image illustrates some of the
transitions in the Balmer series.
|
||||||||||
|
|