Medical Laboratory TechnologyMLAB 1331Laboratory Manual
Laboratory Schedule
LABORATORY EXERCISE 1: Fecal Examination
Part I. Macroscopic Examination
A. Objectives 1. Properly collect a stool specimen for parasitic examination. 2. Correctly describe the consistency and type of parasites found in the different consistencies. 3. Correctly determine the presence of fresh and/or occult blood in a stool specimen.
B. Introduction
The consistency of a stool specimen, whether formed, semi-formed, mushy, or liquid, is of great importance, giving an indication of the types of organisms, which it may contain. Trophozoites are usually found in liquid or soft stools, but almost never in fully formed ones. Cysts are rarely seen in liquid stools, unless as the result of the administration of a cathartic or laxative. Cysts are usually found in fully formed specimens. Helminth eggs may be found in either liquid or formed stools, but as the liquid stool is usually very dilute, they may often be difficult to detect in such specimens.
The surface of the entire specimen should be examined for macroscopic parasites. Frequently pinworms will be seen on the surface of a stool specimen, and tapeworm proglottids may likewise be found there or in the interior of the specimen. The stool should be broken up with applicator sticks to check for helminths. The presence of blood should be noted. Bright red blood on the surface of formed stools is most frequently a sign of bleeding hemorrhoids; bloody mucus in loose or liquid specimens is highly suggestive of amoebic ulcerations in the large intestine, though it may be due to other conditions. Patches of mucus on the surface of a specimen, particularly if blood-tinged, should always be examined with care for trophic amoebae. Occult blood may be present due to parasitic infections, but it is more likely to be indicative of other gastrointestinal disorders. An occult blood exam is often routinely performed on all stools submitted for ova and parasites (O & P) examination.
Freshly passed specimens are essential for the detection of trophic amoebae or flagellates. Therefore, all liquid or soft stools must be examined within one-half hour of the time of passage or preserved in an appropriate manner. The immediate examination of fully formed stool specimens is not so important, but they should be kept under refrigeration or otherwise preserved if examination must be delayed. Fecal specimens should never be incubated at 37°C; this will promote disintegration of trophozoites and cysts and maturation and hatching of hookworm eggs. Ideally, specimens should be preserved if delay in examination is anticipated.
A stool should be collected in a tightly covered, clean (not necessary sterile), container and must not be contaminated with urine. The label on the specimen must include the time of passage of the stool as well as appropriate patient identification information.
C. Procedure 1. Note the color of the specimen. 2. Note the consistency of the specimen. Mushy or liquid stools suggest the possible presence of trophozoites or intestinal protozoa. Protozoan cysts are found most frequently in formed stools. Helminth eggs and larvae may be found in either liquid or formed stools. 3. Examine the surface of the specimen for parasites (e.g., tapeworm proglottids or, less commonly, adult pinworms). 4. Examine the stool for blood and/or mucous. a. Fresh blood (bright red) indicates acute lower intestinal tract bleeding b. Bloody mucus suggests ulceration, and some of this material should be examined microscopically for trophozoites. c. Black, tarry stools are indicative of occult (hidden) blood from higher up in the intestinal tract. 5. Break up the stool with applicator sticks to check for the presence of adult helminths (e.g., Ascaris). 6. Feces should be sieved after drug treatment for tapeworm infections to assure recovery of the scolex.
II. Microscopic Examination
A. Objectives 1. Prepare and examine slides by the direct wet film and iodine stained procedures. 2. State the advantages and disadvantages of direct wet film evaluation.
B. Introduction
The evaluation of saline direct mounts provides opportunity to study the motility of any trophozoites present. While the addition of iodine stains the internal structures necessary for identification of the cysts of some amoeba and other protozoa, trophozoites are rapidly killed and are sometimes unidentifiable. Wet mount procedures should be performed on concentrated specimens.
Recent literature suggests that wet mount preparations on fresh or preserved specimens are of little value, considering the amount of technical time they require. The following direct wet mount procedures are provided for your information since, some laboratories still perform this procedure.
C. Procedures 1. Direct saline wet mount a. Place a drop of saline on the slide.
b. Pick up a small amount of fecal material on the end of an applicator stick. NOTE: Take small amounts of material from several different areas, especially from bloody and/or mucoid areas.
c. Emulsify in the saline and cover with a coverslip. Examine on low and high power. NOTE: A smear should be thin enough so that a printed page can be read through it.
d The entire preparation must be examined for the presence of eggs, larvae and protozoa. Low power is used to scan for large helminth eggs or larvae. High power is used to detect and identify smaller parasites and larger helminth eggs and larvae.
e Any parasites detected are reported out by their scientific name and quantity observed. The following chart describes the method used by the Texas State Health Department for quantitation of parasites found.
If no parasites are observed report out as “No parasites seen.”
2. Iodine Wet mount a. Place a drop of Lugol's iodine solution on a slide. b. Pick up a small amount of fecal material on an applicator stick using the same criteria in the saline procedure for selection of the proper areas. c. Emulsify in the iodine solution and cover with a coverslip. d. Examine on low and high power as described in the previous procedure. NOTES: The film may be made directly with iodine or the iodine may be added to a saline mount by adding a drop to the edge of the coverslip so that it gradually diffuses into the saline mount.
III. Concentration Techniques
A. Objectives 1. Discuss the zinc flotation concentration technique 2. Perform the formalin-ethyl acetate concentration technique 3. Examine the concentrate microscopically using the saline and iodine wet mount procedures.
B. Introduction A fecal concentration technique increases the possibility of detecting parasites when only a few are present in the feces and is a routine part of the clinical examination of feces. Two general types of methods are used – sedimentation and flotation. The sedimentation procedure concentrates parasite stages present in a large amount of feces into about 2 gm. of sediment. The technique involves straining the stool to remove large particles, formalizing to kill and preserve the organisms, and adding a solvent such as ether or ethyl acetate to dissolve the fats. The mixture is then centrifuged and the sediment examined. The zinc-sulfate concentration technique provides a cleaner concentration and is good for the recovery of protozoan cysts and most eggs, but does not work well with trematode eggs or those of the broad fish tapeworm. The flotation method uses liquids of a higher specific gravity than that of eggs and cysts, so that parasites will float to the surface. The top layer is skimmed from the surface and examined microscopically. This procedure must be performed with meticulous attention to detail and is not widely used routinely. Therefore this method will not be practiced in this course.
The following two pages illustrate the appropriate methods for collection and fixing stool specimens.
C. Formalin-Ethyl Acetate Procedure
1. Label a fecal concentration tube (the flat-bottomed container) for each stool to be examined. 2. Add 9 ml 10% formalin to each tube. Using the 5" dispensing spoon, added one rounded spoonful of fresh stool specimen or two spoonfuls of preserved stool specimen. 3. Mix well with the spoon. 4. Add 3 ml ethyl acetate. 5. Take the green strainer and 15 ml conical tube assembly and pull the vent-straw out about 1 inch. Now screw the assembly firmly onto the flat-bottomed tube containing the fecal specimen. 6. Shake vigorously for about 30 seconds. 7. Pointing the conical tube section downwards, shake the specimen through the strainer into the 15 ml conical centrifuge tube. 8. Unscrew the strainer and the flat-bottomed tube and discard. 9. Place the white screw cap on the centrifuge tube. Centrifuge at 2000 rpm for 5 minutes. 10. Using an applicator stick, rim the debris layer. Pour off the debris and supernatant fluid leaving sediment. 11. Examine the sediment using the saline and iodine wet mount procedure. Cysts are refractile and more easily detected unstained and morphology of larval forms is more characteristic.
IV. Preservation of Specimens
A. Objectives 1. State the purpose for using preservatives 2. Compare and contrast the different preservatives used in the laboratory. 3. Properly preserve a fecal specimen and prepare slides for a permanent stained smear.
B. Introduction Trophozoites of intestinal amoebae die and disintegrate rapidly so it is important to achieve adequate and proper preservation when specimens cannot be examined immediately (within 30 minutes of passage). Several preservatives are available. The most popular preservative used by clinical laboratories is Polyvinyl Alcohol-Fixative (PVA-fixative). The purpose of fixatives is to prevent deterioration of fragile forms of parasites. Many commercially prepared kits contain a 2-vial method for patients to submit stool specimens. One vial of the kit contains PVA-fixative and the other contains 5 or 10 percent formalin. It is very important that the proper ratio of fecal specimen and fixative are used. If the patient collects the specimen at home they should be told to fix the specimens immediately after passage. Some organisms rapidly lose their characteristic morphology.
C. Procedures 1. On microscope slides: A drop of dysenteric stool is placed on a microscope slide and mixed with three drops of PVA-fixative. The mixture is then smeared over approximately two-thirds of the glass surface and allowed to dry thoroughly (preferably overnight at room temperature). Dried smears remain satisfactory for extended periods.
2. In Vials: A quantity of specimens is thoroughly mixed in a vial containing three parts of PVA-fixative. Smears for staining can be prepared immediately by spreading a drop or two of the mixture over the surface of a slide. It is important not to have the smears too thick and to allow them to dry thoroughly.
3. Staining of PVA-fixative Preserved Smears: The Trichrome staining procedure is preferred for permanently staining fecal films. It is important that the stained film be protected with mounting medium and a cover slip.
V. Detection of Occult Blood
A. Objectives 1. State the principle of the occult blood including the chemical reaction involved. 2. Explain dietary restrictions recommended to patients prior to collection of occult blood specimens, including why certain foods should be avoided. 3. Properly perform and interpret the occult blood test.
B. Introduction As blood passes through the intestinal tract it changes color and consistency due to the digestive processes. Stools with large amounts of occult blood are black and have a tarry consistency. Smaller amounts of blood may not be visibly detectable. Occult blood may indicate lesions in the intestinal tract due to parasitic infections, cancerous tumors, bleeding ulcers or other disease processes. The occult blood test is primarily done to screen for colo-rectal cancer. It is an easy inexpensive procedure and the specimen is easily obtained by the patient. The chemical reaction involved is as follows: Hemoglobin + Developer Hb + 2 H2O2 ® 2H2O + O2 Oxidation of Guaiac O2 + Guaiac ® Oxidized Guaiac (Colorless) (Blue) The presence of a blue color upon completion of the test is a positive test. A positive and negative control must be run concurrently with the patient sample. Most manufacturers now include this as part of the patient test card.
It is recommended that the patient be placed on a high residue diet starting 2 days before and continuing through the test period.
DIET MAY INCLUDE: 1. Meats: Only small amounts of chicken, turkey and tuna. 2. Vegetables: Generous amounts of both raw and cooked vegetables including lettuce, corn, spinach, carrots and celery. Avoid those with high peroxidase activity. 3. Fruits: Plenty of fruits, especially prunes and apples. 4. Cereals: Bran and bran-containing cereals. 5. Moderate amounts of peanuts and popcorn daily. If any of the above foods are known to cause discomfort, the patient is instructed to inform his physician. TO BE AVOIDED: 1. Meat: Diet should not include any red or rare meat. 2. Fruits and vegetables containing high peroxidase activity: Turnip Cauliflower Broccoli Cantaloupe Horseradish Parsnip Alternately, the special diet may be omitted initially with dietary restrictions imposed upon the re-testing of all positive results.
Other factors which affect the test: 1. Medications: Do not ingest aspirin, tonics or vitamin preparations which contain Vitamin C (ascorbic acid) in excess of 250 mg per day. 2. Bleeding hemorrhoids 3. Collection of specimen during menstrual cycle. 4. Improper specimen collection 5. Other diseases of the gastrointestinal tract such as colitis, gastritis, diverticulitis and bleeding ulcers.
Specimen Handling: It is important that the stool specimen be applied as a very thin smear to the ColoScreen Slides. The smears may be prepared and developed immediately or prepared and stored up to 12 days prior to development. Care should be taken so that anything coming into contact with the specimen is free of blood. Because of the non-homogeneity of the stool, it is recommended that the test be performed on three (3) consecutive evacuations.2,3
Interfering Substances: Ingestion of ascorbic acid (Vitamin C) in high doses has been shown to cause false negative results and intake should be discontinued prior to and during the test period. Peroxidase from fruit and vegetables can cause false positive results. Elimination of red meat from the diet during the test period eliminates the source of hemoglobin, which can cause false positives. Any oral medications, which may cause irritation of the gastrointestinal tract and possible occult bleeding, should be discontinued during testing (i.e., aspirin, indomethacin, etc.).
C. Procedure
1. Supply all information requested on the front flap of the Slide. 2. Open the front flap. 3. Collect a small amount of stool specimen on one end of applicator. Apply a very thin smear in Box A. 4. Reuse applicator to obtain a second and a third sample from different parts of the stool specimen. Apply a very thin smear inside Box B and Box C respectively. (On subsequent bowel movements, repeat above steps on additional slides.) 5. Close the cover. 6. Open perforated window on the back of the slide. 7. Apply two (2) drops of Developer to the backside of boxes A, B and C. 8. Read results after 30 seconds and within 2 minutes. 9. Record the results; any trace of blue color, within or on the outer rim of the specimen is positive for occult blood.
VI. Calibration of Ocular Micrometer
A. Objectives 1. Calibrate the ocular micrometer to measure parasites. 2. Measure parasites using the micrometer to accurately determine their size.
B. Introduction Size is one of the most important criteria used in identifying parasites or their diagnostic stages. The unit of measurement is the micromillimeter (μm), sometimes referred to as a micron(μ). A rough estimation can be obtained by comparing the parasite with the size of human red blood cells (7.5 to 8μ). However, if an organism is to be accurately measured, a calibrated micrometer disc placed within the ocular of the microscope should be used. The type suggested is a disc with a line scale divided into 50 spaces or ocular micrometer units. Ocular micrometers are relatively inexpensive and are available through several scientific supply companies.
Inasmuch as the units on the ocular micrometer are arbitrary and the exact value of each varies with different objectives and microscopes, the value must be calculated with each combination of lenses – low, high-dry, and oil-immersion. To do this, the ocular micrometer units are compared with a scale of known dimensions. This may be done by superimposing the image of the unknown ocular scale on the known scale of a stage micrometer (slide with a carefully etched and calibrated scale of 0.1-mm and 0.01-mm divisions) as demonstrated in figure 2. Stage micrometers are considerably more expensive than ocular micrometers and unless microscopes are calibrated frequently, a stage micrometer might be borrowed rather than purchased. Several State Health Department Laboratories have stage micrometers for loan; CDC also has some available for 10-day loan periods.
C. Procedure
Caution, all materials used must be clean and lint free.
1. Remove the 10X ocular from the microscope and unscrew the top or bottom lens, depending on the model of microscope. Place the micrometer disc on the diaphragm within the ocular or on top of the bottom lens in newer models so that the engraved side is underneath (fig.1). Screw back the lens and insert the ocular in the microscope. Be careful to keep both micrometer and lens clean and free of dust, which might interfere with observations.
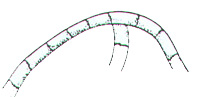
2. Place the stage micrometer on the stage and focus on some portion of the scale with the 10X objective.
3. Looking through the microscope, examine the ruling of the stage micrometer so that you can distinguish between the large (0.1-mm) and the small (0.01-mm) divisions.
Ocular Micrometer – Top Scale Stage Micrometer – Bottom Scale
4. Adjust the field so that the 0 line on the ocular micrometer is exactly superimposed upon the 0 line on the stage micrometer. If the two scales are placed so the lines intermesh but the scale numbers are apart, readings are easier.
5. Without moving the stage micrometer, find another point at the extreme right where two other lines are exactly superimposed. The second set of superimposed lines should be as far as possible from the 0 lines, but the distance will vary with the objective and microscope.
6. Knowing that each large division of the stage micrometer equals 0.1 mm and each small division equals 0.01 mm, determine the total distance (in millimeters) between the two points of superimposition and the number of small ocular units necessary to cover the same distance (fig. 2). Example: Suppose 49 ocular units equal 8 large stage units or 0.8 mm.
7. Calculate the number of millimeters that is measured by one small ocular unit. 8. Because measurements of small organisms or structures are usually given in micromillimeters (μm), or microns (μ), rather than millimeters, the above determination must be converted to micromillimeters. Since 1 mm equals 1,000 μm, the millimeter determination multiplied by 1,000 will give the number of micromillimeters measured by 1 small ocular unit.
Example: 0.0163 mm X 1,000 = 16.3μm
9. Record the calibrations of the ocular micrometer obtained with the 10X oculars combined with each of the three objectives. The lines of the stage micrometer will increase in magnification while those of the ocular micrometer will remain the same. With high and oil magnifications, the thinner ocular micrometer line must be centered on the broader stage micrometer line for more accurate measurement. 10. The size of any microscopic object can be quickly determined by measuring it with the ocular micrometer, noting the particular lens combination, and referring to the record of the ocular micrometer calibrations. For ready reference, the values for 1 to 50 units can be recorded in a chart such as the one following.
Ocular Micrometer Calibrations Ocular Magnification 10X
VII. References
A. Medical Parasitology, A Self-Instructional Text, Leventhal, Ruth, pp. 123-132. B. Laboratory Confirmation of Intestinal Parasites, Workshop Manual, Texas Department of Health pp. 13-14, 17-20 C. Coloscreen, Reagent Package insert
LABORATORY EXERCISE 1: Fecal Examination Worksheet
I. Color
Appearance
II. Direct smear
saline
iodine
III. Formalin-ethyl acetate Concentrate
saline
iodine
IV. Occult blood
positive control
negative control
patient
V. Ocular micrometer calibration
low power objective
high power objective
oil immersion objective
microscope number
LABORATORY EXERCISE 2: Permanent Stained Smears
A. Objectives 1. Compare and contrast the iron-hematoxylin and trichrome stains 2. Correctly perform the trichrome stain on fecal material 3. Using appropriate technique, examine permanently stained smears, including acid-fast for cryptosporidium.
B. Introduction It is now routine procedure for laboratories to perform permanent stained smears on all fecal specimens submitted for ova and parasite (O & P) examination. Such preparations facilitate detection and identification of trophozoites and cysts by improving the cytological detail of the organisms. Stained smears also provide a permanent record of organisms encountered and can also be used for teaching purposes for years. Furthermore, small organisms may be found on stained films, which are missed by other examinations. The classical method is the long Heidenhain's iron-hematoxylin method, but for routine diagnosis, shorter procedures such as Wheatley's trichrome method, have been developed.
C. Procedures Iron-hematoxylin Stain Though the iron-hematoxylin stain yields better results in cytological detail, this stain requires considerable technical competence, and will not be practiced in this course.
Trichrome Technique for Staining Intestinal Protozoa The trichrome technique of Wheatley is a rapid staining procedure, which gives good results for routine purposes. The method is simple in that overstaining and differentiation are not necessary to bring out the morphologic details of the parasites nor is it necessary to mordant before staining. The stain solution is stable and may be used repeatedly, the lost volume being replaced by the addition of stock solution. Staining over 15 smears daily (in 50 ml. of stain), however, tends to weaken the stain. Strength will return upon standing, if stain is allowed to evaporate in open air for 3 to 8 hours.
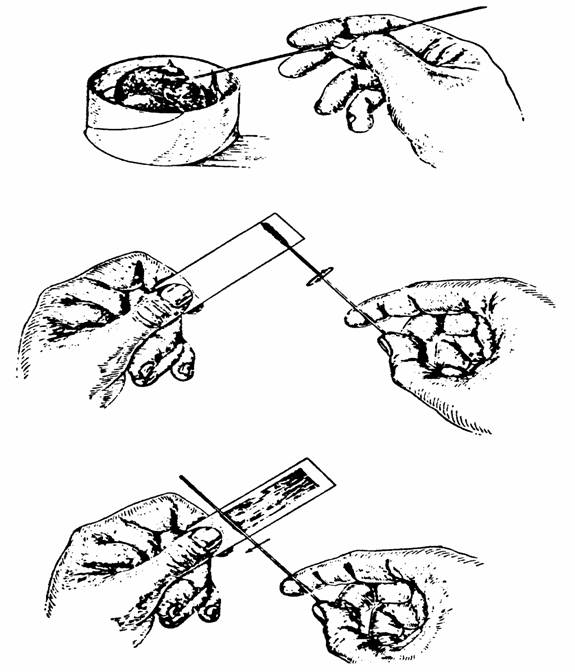
Preparation of fecal film for permanent staining.
The smear is prepared from either fresh or preserved fecal material using the technique shown by the following diagram.
The staining of fresh and PVA-fixed material differs chiefly in the increased time required for the latter and the omission of the fixative step since the material in the PVA solution is already fixed.
Staining Procedure with Fresh Specimens
Staining Procedure with PVA Films
Stain Reactions (Trichrome)
Thoroughly fixed and well-stained E. histolytica cysts are blue-green tinged with purple; E. coli cysts, slightly more purplish. Background material usually stains green, resulting in a noticeable color contrast with the protozoa. Organisms in thick smears take the more neutral shades of red and green. In contrast with those stained with hematoxylin, such smears have a transparency, which enables identification of imbedded protozoa.
Protozoa and eggs are less subject to distortion, however, in thin smears. Eggs and larvae usually stain red and contrast strongly with green background. Thin-shelled eggs usually collapse when placed in mounting medium although some diagnostic features may be retained especially if smear is examined immediately.
Large protozoa are readily picked up with a low power objective, using 10X ocular. Smaller forms, (E. hartmanni, E. nana, etc.) and those staining faintly, are more visible with high dry and oil immersion lenses. A 43X or 44X oil immersion objective is useful in examining the smears.
Non-staining cysts and those staining predominantly red are most frequently associated with incomplete fixation. If unsatisfactorily stained organisms are obtained from specimens submitted in PVA-fixative, it usually indicates incomplete fixation associated with poor emulsification. Thorough emulsification of preferably soft stools will yield critically stained cysts and trophozoites. Degenerate forms stain pale green.
Mononuclear and polymorphonuclear leukocytes as well as Blastocytis hominis present the same diagnostic problems as when stained with hematoxylin. The cytoplasm of pus and tissue cells, however, does stain more greenish.
Suggestions for Examination of Stained Fecal Smears
Stained smears should be examined for organisms only with oil immersion. High, dry examinations are of little value and may lead to erroneous diagnosis. However, low power (10X objective) can be used to locate appropriate areas of the smear to be examined. A suggested procedure follows.
With low power, scan the smear for light and dark (thin and thick) areas. In a heavier smear, better-stained organisms will usually be found in the thinner areas; and in lighter preparations, they are usually more easily detected and identified in the thicker areas. Sometimes, it is advisable to examine both areas since different species of organisms vary in stain intensity. Also, because organisms are not uniformly distributed in the feces, different parts of the smear should be examined. We recommend looking at fields in the upper, center, and lower areas or in the left, center, and right areas of the smear.
After locating an appropriate area, switch to oil immersion for examination.
If the selected area appears to be too light or too dark under the oil, try another spot. There is little advantage in examining areas that are much too light or too dark.
An experienced technologist usually can adequately examine a stained smear in about 15 to 20 minutes. Occasionally, problem smears or problem organisms will take longer.
The species and stage of organism will influence the stain reaction. In a well-stained smear, some species will usually stain lighter than others, and trophozoites will stain lighter than cysts.
Species which usually stain distinctly and medium to deep in intensity:
E. coli cysts and trophozoites E. histolytica cysts and trophozoites I. butschlii trophozoites and cysts E. nana trophozoites (usually lighter than those of I. butschlii) G. lamblia cysts, occasionally C. mesnili cysts, occasionally
Species which tend to stain light to medium in intensity:
E. nana cysts (sometimes very light) D. fragilis trophozoites (light to medium) E. hartmanni cysts and trophozoites (vary in intensity) C. mesnili trophozoites (often very light) G. lamblia trophozoites (vary, but usually are light) G. lamblia & C. mesnili cysts, occasionally (more often these are medium)
Name________________________________
Date_ _______________________________
LABORATORY EXERCISE 2: Permanent Stained Smears Worksheet
I. Trichrome Stain
II. Acid Fast
LABORATORY EXERCISE 3: Identification of Parasites
A. Objectives
1. Observe parasites in known specimens and study characteristic morphology. 2. Correctly identify unknown organisms in formalin and trichrome preparations. 3. Observe and study parasites in blood, tissue and other samples as available.
B. Introduction
In the previous laboratory exercises students were introduced to the basic methods used to detect, quantitate and measure parasitic forms. These skills will now be put to use to detect and study parasites in known specimens. Once the student has sufficiently studied the known specimens, unknown specimens will be provided by the instructor for identification. The instructor will also set up the teaching scope to allow students to study certain specimens which are available in a limited supply. These will be studied as knowns and again later as unknowns.
C. Procedure 1. Morphologic characteristics of parasites are covered in the lecture component. The organisms covered in lecture should then be studied by the student in the laboratory component. Use the charts found in this laboratory exercise along with your textbook to aid in identification of the known and unknown specimens. Have the instructor verify your organism and check off the organism on your recording results sheet.
2. Upon completion of the known organisms of the group being studied request unknowns from the instructor. Upon successful identification have instructor check off on the recording results form.
3. A student will have two opportunities to identify each unknown. In specimens with more than one organism, the student will be allowed two opportunities for each organism.
4. Students will be awarded points for each successful identification according to the list on the recording results form.
Name Date
LABORATORY EXERCISE 3: Known Specimens Worksheet
Name
LABORATORY EXERCISE 3: Unknown Specimens Worksheet (100 points) F = Formalin preserved specimen T = Trichrome smear O = Other
Each student will be provided 25 unknown specimens to identify. Specimens are worth 4 points each. For each unknown, you are given two (2) chances to correctly identify the organism(s). If you are completely correct on the first attempt, the full credit will be awarded; if however, you are not correct the first time, but are correct the second time, one (1) POINT will be deducted. Extra credits may be given, at the instructor’s discretion, for additional organisms found in unknown specimens. Each student is expected to work independently when identifying his or her unknown. After identifying the organism and marking your paper, leave it under the microscope while you verify its identity with the instructor. You may be asked to show the instructor the organism.
The instructor will hand out individual unknown report sheets
LABORATORY EXERCISE 4: Scotch Tape Preparation
A. Objectives 1. State the proper method for performing the scotch tape preparation. 2. Identify parasites present on a scotch tape preparation
B. Introduction The female pinworm (Enterobius vermicularis) migrates out of the anus at night to deposit her eggs on the perianal region. These eggs are easily recovered by pressing a piece of scotch tape against the perianal region. The tape is then placed on a microscope slide and submitted to the laboratory for examination. Parents can collect the specimen from a young child at home using a kit supplied by the laboratory. The specimen should be obtained first thing in the morning before the patient bathes or defecates.
C. Procedure 1. Fold the edges of a piece of clear (not frosted) cellophane tape around the edge of a tongue depressor so that the sticky side faces out. 2. Spread buttocks and apply tape to the anal area using a rocking motion to cover as much as the perianal mucosa as possible. 3. Remove the tape and apply it to a microscope slide, sticky side down. Press firmly so that no air bubbles are trapped. 4. Examine the slide for pinworm eggs under low power using decreased light. Be sure to examine the entire area under the tape. The eggs are colorless, therefore, good focus and low light are critical. NOTE: Pinworm infection should not be ruled out until at least five daily consecutive specimens are negative.
LABORATORY EXERCISE 5: Preparation and Examination of Blood Films
A. Objectives 1. List two organisms which may be detected in fresh blood specimens. 2. Correctly prepare a thin blood smear. 3. Correctly prepare thick blood smears for parasitological study. 4. State the proper technique for examination of thin and thick blood smears.
B. Introduction
The confirmatory test for diagnosis of blood parasites is made through the examination of fresh blood and preparation of blood smears which are then examined for the presence of the parasites. Examinations of blood smears are very time consuming. The parasite may be present in the peripheral blood in very small numbers. Careful examination of the blood smear is required before reporting the smear out as “no parasites seen.” The presence of even one parasite on a blood smear is significant and should be species identified if at all possible.
It takes experience to accurately examine blood smears for parasites. Platelets sitting on top of red blood cells are commonly mistaken for blood parasites. Any suspicious looking organisms should be confirmed by fellow technologist and then the pathologist.
C. Procedures
1. Examination of Fresh Blood
Microscopic examination of fresh blood is not undertaken routinely but is useful for the detection of trypanosomes and microfilariae, which may be easily recognized by their characteristic motility in fresh blood. A small drop of blood is placed on a slide and covered with a coverglass to prevent clotting. The blood is examined under low power for whip-like motions of microfiliariae and the rapid undulating and twisting movements of trypanosomes.
2. Thin Film
Thin blood films are used for the specific identification of malarial parasites, trypanosomes, and microfilariae. It is essential that the film be really thin. The chief advantage of a thin film is that it preserves the structure of the parasites with a minimum of distortion.
The thin films are made in the same way smears are made for hematological study. 3. Thick Film
The thick film is preferred over the thin film for diagnosis of malaria because it reveals, comparatively quickly, sparse or scanty infections such as occur in new or chronic cases. It has been estimated by various workers that the thick film enables one to examine 10 to 50 times as much blood as the thin films in the same length of time.
a. Place a large drop of blood about one inch from the end of a clean glass slide. With the corner of another clean slide spread this blood to the size of a dime, using a circular motion and rotating through the blood approximately 20 or 40 times to thoroughly defibrinate it. See diagram at right.
b. Two thick or a thick and a thin may be put on the same slide. The blood is then allowed to dry while the slide in on a level surface. After several hours of drying, the films are ready to staining.
c. Blood films are usually prepared when the patient is admitted; samples should then be taken at intervals of 6 to 18 hours for at least 3 successive days.
d. If fresh capillary blood is not available, request a tube of fresh EDTA blood and prepare smears immediately after the blood is received.
D. Staining Smears for Malaria
1. Blood films should be stained as soon as possible, since prolonged storage results in stain retention. 2. The stains that are generally used are of two types. a. Has the fixative in combination with the staining solution (Wright's stain). b. Has the fixative and stain separate (Giemsa's stain).
E. Interpretation of Smears for Malaria
The thick film is used for the detection of the presence of malaria parasites. The thin film is used primarily for specific parasite identification. Generally the pathologist should aid in the identification of any parasites detected.
A minimum of 100 microscopic fields should be examined before a film is reported out as negative. LABORATORY EXERCISE 6: Plating Fungal Cultures
A. Objective
1. To subculture three species of fungi to Sab-dex slants utilizing aseptic technique. 2. Determine the growth rate of the fungi.
B. Introduction
When culturing clinical material for fungi or yeasts, two tubes or plates of media should always be used. Both tubes should be nutritionally rich, but one should contain antibiotics (usually cycloheximide and chloramphenicol). Tubed media are preferred over plates, as the former will not dry out over the long incubation period, and also the chance for fungal reproductive structures to become airborne and contaminate the laboratory and personnel is decreased. Never use plates when Coccidioides immitus is suspected; this fungus is extremely infectious and aerosols may be inhaled.
Always work under a microbiologic hood, wear gloves, autoclave specimens and inoculated media when finished, and disinfect the work area daily.
As a general rule, there should be minimal time between collection of the clinical specimen and culture; avoid refrigeration of the specimen if possible. If media has been refrigerated, remove it from the refrigerator and allow it to warm to room temperature prior to using it.
Soft or liquid material can be inoculated directly to the surface of the media by using a regular bacteriologic loop, sterile applicator stick or sterile Pasteur pipet. Hard specimens such as nails and skin require imbedding the material into the agar using tweezers or a probe.
When subculturing cultures to fresh slants, use a loop or sterile applicator stick to recover spores from the surface of the culture and apply them directly to the surface of the fresh slant.
During this laboratory session the cultures used have been grown sterile stock cultures and should contain only one organism. Experience will be gained in the appropriate method of transferring the stock culture to sterile media using appropriate aseptic technique.
C. Materials
1. Sterile applicator sticks 2. One tube of Sab-dex agar for each fungi to be subcultured 3. Incinerator
D. Procedure
1. Properly label a fresh tube of Sab-dex for each fungi to be subcultured (name of organism, date and your initials). 2. Loosen the cap on the fresh tube of agar and place in test tube rack. 3. Carefully remove cap on the tube of the fungi to be sub-cultured. Hold the open end of the tube in front of the incinerator for a few seconds, flame the loop, place it in the culture and cool it in the agar, carefully collect the fungal specimen by gently scraping the top of the culture (the tallest portion of the fungi contain the reproductive structures of the fungi) or, if the culture is a yeast, scrape a small portion of the culture from the outermost section (this is the youngest portion of the culture). 4. Flame the open end of the culture tube again and carefully replace the cap. Immediately remove the cap on the fresh media, flame the opening, and carefully inoculate the media with the material on the loop. 5. Flame the open end of the culture tube again, replace the lid leaving it fairly loose, and immediately flame the loop. 6. Repeat this process for each mycology culture to be subcultured. 7. Incubate cultures at room temperature. LABORATORY EXERCISE 7: Macroscopic Colonial Morphology
A. Objectives
1. Utilize the appropriate terms to describe the colonial morphologic characteristics of fungal cultures regarding the texture, topography and color of the cultures. 2. Differentiate molds from yeasts based on macroscopic morphology.
B. Introduction
Fungi is a term that includes both yeasts and molds. The fungi seen in the clinical laboratory can easily be separated into two groups based on the macroscopic appearance of the colonies formed. The yeasts produce moist, creamy, opaque or pasty colonies on culture media, while the molds produce fluffy, cottony, woolly or powdery colonies.
Mycology cultures should be observed daily (or at least every other day) for growth. If a fungus is detected, its rate of the growth is noted, and its morphology must be described to aid in the identification.
The colonial texture describes the height of the aerial hyphae above the agar surface. Colonial topography describes the various designs of hills and valleys seen on fungal cultures. The topography is often masked by the aerial hyphae; therefore, this characteristic is better observed on the reverse side of the colony. A colony may possess no topography, that is, it is flat.
The last characteristic of macroscopic morphology is the color of the colony. Be as specific about the colony colors as possible. For example, instead of describing a culture as brown, use words such as beige, tan, khaki, or mahogany. If there are concentric rings of different colors describe each one. Be sure to characterize both the front and reverse sides of the culture.
C. Materials - Stock cultures
D. Procedure
1. Label a sheet of legal size paper with the following headings: a) name of culture, b) rate of growth, c) texture, d) topography, e) front color and f) reverse color. 2. Use the following terms to describe the growth rate: a) Rapid growth rate - forming a mature colony within five days. b) Intermediate growth rate - forming a mature colony within 6 to 10 days. c) Slow growth rate - forming a mature colony in 11 to 21 days. 3. The following terms are commonly used to describe colonial texture, you may find other appropriate terms throughout your textbook: cottony, velvety, granular and glabrous. 4. The following terms are commonly used to describe colonial topography, your may find other appropriate terms throughout your textbook: rugose, umbonate and verrucose. 5. Describe both the front and reverse sides of the culture. EXAMPLES OF COLONIAL TOPOGRAPHY
LABORATORY EXERCISE 8: Germ Tube Test
A. Objective
1. Correctly perform and interpret the germ tube test.
B. Introduction
This is a test for the presumptive identification of Candida albicans. Strains of C. albicans produce germ tubes from their yeast cells when placed in a liquid nutrient environment and incubate at 35C for 3 hours (similar to the in vivo state). Not all C. albicans isolates form germ tubes, especially those from cancer patients on therapy or persons on anti-Candida antibiotics. Therefore this test must be combined with other procedures for definitive identification.
C. Materials
1. Media Preparation a. Place 0.5 ml of human serum into appropriately labeled tubes.
2. Yeast cultures
D. Procedure
1. Inoculate the tube of serum with a small amount of the young test organism. Too large an inoculum will inhibit germ tube formation. Be sure to set up positive and negative controls, particularly if human serum is to be used, to assure that the serum does not possess anti-Candida antibodies or other inhibitory factors. 2. Incubate the tubes at 37C for 3 hours. Note: The test can be examined after one-hour incubation. Many clinical sites utilize a one-hour incubation time. 3. Place a drop of the suspension on a slide, put on a coverslip, and examine it microscopically for long tube like projections (germ tubes) extending out from the yeast cells. 4. Draw a picture of the yeast cells and/or germ tubes you observed in each tube. Turn it in for credit for this lab.
E. Interpretation
The germ tube is a short hypha outgrowth from the yeast cell, has parallel walls at its point of origin and is aseptate. That is, there is no constriction at the point of outgrowth. Be careful not to confuse germ tubes with pseudohyphae which may not necessarily have parallel sides, may be septate and have constrictions. Refer to your textbook for additional information. LABORATORY EXERCISE 9: Slide Culture
A. Objective
1. To properly prepare slide cultures from stock cultures utilizing appropriate aseptic technique. 2. Observe and record the microscopic morphology of the slide cultures.
B. Introduction
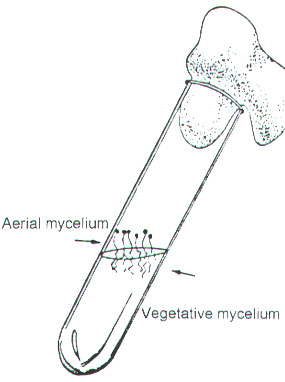
The slide culture technique has been a tool in mycology studies for many years and is performed with many modification in laboratories worldwide.
The advantages of a slide culture is that the fungal elements are grown and maintained in their original juxtaposition, thus making it easier to morphologically identify the organism. When properly prepared conidia will be observed still attached to their condiogenous structures which is of critical importance in their identification. Two mounts are obtained from one culture.
The disadvantages of this method are that it requires some technical expertise to set up the slide culture, and there is a waiting period for incubation of the slide culture in addition to incubation on the primary isolation media. Cottony fungi may grow past the edges of the coverslip before forming reproductive structures.
C. Materials
1. Medium preparation a. Melt a tube of potato dextrose agar. b. Pour melted agar into petri dish. Cover and allow to cool. c. Cut the agar into small cubes (smaller than a cover slip).
2. Sterile applicator sticks 3. Sterile slides and coverslips 4. Sterile petri dishes 5. Cotton balls 6. Incinerators
D. Procedure
1. Bind together the ends of two applicator sticks with rubber bands. Break the sticks almost all the way through the middle so they form a V-shape. The sticks will serve as a platform on which to place the slide culture. 2. Place the sticks in alcohol, dry and put in the bottom of a sterile petri dish. 3. Label a glass slide, dip in alcohol, flame dry, and set across the platform. 4. Place 1 square block of the potato dextrose agar (PDA) on the slide. This particular medium greatly enhances sporulation, although occasionally Sabouraud dextrose agar works better. 5. Using aseptic technique, inoculate a small piece of fungal colony from the primary isolation tube to one side of the agar block. When obtaining the inoculum, be sure to cut into the colony to get the vegetative as well as aerial growth. Do not take inoculum from the center or edge of the colony. Repeat this procedure for each side of the agar block. 6. Dip a glass coverslip (22x40 is recommended) in alcohol, flame, and place over the inoculated agar block. Press down lightly to ensure good contact between the agar and coverslip. 7. For moisture, pour about 5 ml of sterile distilled water into the bottom of the Petri dish. Cover the dish and set at room temperature to incubate. If all the water evaporates with time, add more. 8. The fungus will first grow on the sides of the agar, then out onto the slide and coverslip. Periodically examine the slide culture under a microscope. Look for fungal maturation, that is, characteristic reproductive structures. 9. When the maturation is evident, remove the coverslip from the slide culture. Usually the agar block will come off with the coverslip; with a teasing needle, gently loosen the suction between agar and coverslip and decant the agar into the biohazardous waste bag. Place the coverslip on a clean slide with one drop of LPCB. 10. If still remaining, remove the agar block from the bottom slide and put it in the biohazardous waste bag. Put a drop of LPCB on this bottom slide and add a clean coverslip. DO NOT TAMP DOWN THE COVERSLIP, as this may jar the conidia loose from the hyphae. 11. Observe the LPCB mounts under a microscope. 12. Use the slide cultures to continue drawing the microscopic fungal morphology. 13. When your have finished drawing the microscopic morphology, place the properly labeled slide on the counter in the mycology area so other students may use it.
LABORATORY EXERCISE 10: Microscopic Morphology of Fungal Cultures by Tease Preparation
A. Objectives
1. Prepare tease preparations of stock cultures. 2. Observe and record the microscopic morphology of fungal cultures.
B. Introduction
Tease mounts are used primarily for fungi. Wet mounts are appropriate for yeast-like organisms. The primary purpose of preparing a tease mount is to demonstrate conidia or other reproductive structures or morphological forms which might give information toward the identification of the organism. The definitive identification is based on the characteristic shape, method of production and arrangement of the conidia; however, the size of the hyphae also provides helpful information. The large ribbon like hyphae of the zygomycetes are easily recognized, while small hyphae, 1 to 2um in size, may suggest the presence of one of the dimorphic fungi.
The fungi may be prepared for microscopic observation using several techniques. The procedure traditionally used by most laboratories is the tease mount preparation. This can be done easily and quickly and is often sufficient to identify many of the fungi commonly encountered in the clinical laboratory. A major disadvantage of the tease prep is that the characteristic arrangement of conidia may be disrupted when pressure is applied to the coverslip. If this cannot be avoided, it may be necessary to set up a slide culture.
The easiest, most economical and suitable method for the microscopic identification of fungi is the Scotch tap preparation. The transparent tape preparation allows the observation of the organism microscopically as it has grown in culture. The conidia are usually intact, and the microscopic identification of an organism can be made with ease. However, if the tape is not pressed firmly enough to the colonies' surface, the sample may not be adequate for an identification.
C. Materials
1. Wire bent at a 90-degree angle. 2. Bacteriologic loop holder 3. Incinerators 4. Slides and coverslips 5. Lactophenol cotton blue (LPCB) 6. Clear cellophane tape 7. Teasing needles
D. Procedure
1. Tease Prep
a. Put one drop of LPCB stain on a clean glass slide. b. With a flamed and cooled bent wire pick up a small portion of the fungal colony, cutting through the aerial and vegetative mycelium. Avoid taking the center or the outer edge of the colony; the center is so old that hyphae may be sterile, while the edge is so young that reproductive structures may not yet have formed. c. Place the fungal portion in the LPCB and, with a second teasing needle, tease apart the hyphae so that they form a thin layer. d. Apply the coverslip and carefully press down hard to spread out the fungus. Examine under the microscope for reproductive structures. e. A permanent preparation may be made by rimming the edges of the coverslip with clear nail polish or Permount. f. Label the slide and after examining and drawing the microscopic morphology place the slide in the Mycology area for use by fellow students.
2. Scotch or Cellophane Tape Preparation
a. Put one drop of LPCB on a properly labeled glass slide. b. Touch the adhesive side of a small length of transparent tape to the surface of the colony. c. Adhere the length of tape to the surface of the microscope slide. d. Observe microscopically for the characteristic shape and arrangement of the conidia. Draw the structures on the appropriate area of your paper. When finished place the slide in the Mycology area for use by fellow students.
E. Note
Use your textbook or other reference when looking for the characteristic morphologic features of the culture you are observing. It is important to see how conidia are borne on the hyphae to aid in the identification of the fungus. If only free conidia are observed make another slide using care in selection of the appropriate area for sampling. The best mounts to observe are those obtained by the slide culture technique.
Microscopic Description
LABORATORY EXERCISE 11: India Ink Preparation
A. Objective
1. Properly perform and interpret the India ink preparation. 2. Discuss the limitations of the India ink procedure and state why it is not the test of choice for identification of Cryptococcus neoformans.
B. Introduction
Traditionally, the India ink preparation has been the most widely used method for the rapid detection of C. neoformans. An evaluation of 39 consecutive patients with cryptococcal meningitis seen at Mayo clinic showed that only 40% gave positive India ink preparation of the cerebrospinal fluid.
The purpose of the India ink preparation is to delineate the large capsule of C. neoformans, since the ink particles cannot penetrate the capsular polysaccharide material.
Because of the low rate of positive identifications using the India ink preparation, it is not recommended as a routine tool in the clinical microbiology laboratory. It should be replaced with the cryptococcal latex test for antigen. Some clinical laboratories still perform this test. If a positive is observed the laboratory must perform other studies, such as dark brown pigment production on caffeic acid agar or specific carbohydrate assimilation reactions, before calling an organism C. neoformans. A positive latex test is definitive for the diagnosis of C. neoformans.
C. Materials
1. India ink or nigrosin 2. Microscope slide and coverslip 3. Stock cultures 4. Inoculating loop 5. Incinerator
D. Procedure
1. On a slide, mix a tiny drop of India ink or nigrosin with one drop of specimen sediment, add a coverslip, and let the mount sit 10 minutes to allow yeast cells to settle into one plane of focus.
2. Observe the preparation under the microscope, with the condenser adjusted for maximum light, for capsules around budding yeast. 3. Perform this procedure on the Cryptococcus albidus and Candida albicans specimens. Draw a picture on a separate sheet of paper and turn in for credit for this lab.
* SDA - Sabouraud Dextrose Agar
SDAcc - Sabouraud Dextrose Agar with Cycloheximide and Chloramphenicol. (SDAcc is inhibitory to most of these fungi, however, some strains may grow fairly well in the presence of these antibiotics.)
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||